Athors: Veroniki F1 MD, PhD, Ourailoglou V2 MD, PhD, Fyntanidou B2 MD, PhD, Tsotsolis N3 MD, PhD, Kotso E4 MD, Kyparissa M2 MD, PhD, Grosomanidis V2 MD, PhD
1Anesthesiology Department, “O Agios Dimitrios” General Hospital, Thessaloniki, Greece
2Anesthesia and ICU Clinic AHEPA University Hospital, Thessaloniki, Greece
3Anesthesiology Department, “Saint Luke” Private Clinic, Thessaloniki, Greece
4Intensive care Unit Papageorgiou General Hospital, Thessaloniki, Greece
ABSTRACT
The occurrence of intraoperative oxygenation impairment is common even in healthy individuals and will vary depending on the patient and the type of surgery. The purpose of this study was to evaluate the incidence of oxygenation impairment in patients with normal lung function and to study the safety and efficacy of three different recruitment strategies. Out of a total number of 430 patients, 150 patients developed intraoperative oxygenation impairment, which was defined as the drop of PaO2/FiO2 ratio below 300. These 150 patients were randomly assigned into four study groups according to the recruitment strategy applied. Group A (N: 38): Application of three hyperinflations of the lungs at airway pressure 40cmH2O for 10 sec, followed by an increase in PEEP from 5 to 10cmH2O. Group B (N: 38): Increase in PEEP from 5 to 10cmH2O. Group C (N: 37): Application of three hyperinflations of the lungs at airway pressure 40cmH2O for 10 sec, without any PEEP alteration and Group D (N: 37): No maneuver. Measurements were taken at the phase of oxygenation deterioration and at 5, 15, 30, 45 and 60min after applying the maneuvers and also before extubation. From a total of 430 patients 150 developed oxygenation impairment (38.4%). The median onset time of the deterioration was 30min after intubation and mechanical ventilation. In group A the PaO2/FiO2 ratio increased significantly immediately after the maneuvers and remained elevated until extubation. In group B the PaO2/FiO2 ratio presented a gradual increase to significant levels before extubation. In group C, the instant post-maneuver increase of oxygenation was not sustained until extubation. Finally in group D a gradual decrease of the PaO2/FiO2 ratio was recorded until the end of surgery. According to the results of our study, one third of patients developed intraoperative oxygenation impairment approximately half an hour after intubation. The application of three hyperinflations of the lungs at an airway pressure of 40cmH2O for 10 sec, followed by an increase in PEEP from 5 to 10cmH2O proved to be the most effective treatment of impaired oxygenation.
INTRODUCTION
Impaired oxygenation during general anesthesia is a frequent finding and has been recognized as a clinical entity several decades ago1,2.
It has been estimated that almost 50% of the patients undergoing elective surgery will develop some kind of impaired oxygenation intraoperatively. In most cases intraoperative hypoxemia is mild to moderate and does not cause any problems to the patients1. However, in cases of preexisting perfusion disturbances, hypoxemia could lead to dangerous ischemia. Moreover, intraoperative events of impaired oxygenation seem to predispose to postoperative pulmonary complications3,4.
Induction of anesthesia itself, placement of the patient in the Trendelenburg, forced lithotomy or lateral position, positive pressure mechanical ventilation and patients’ co-morbidities are some of the factors affecting the respiratory system and contributing to intraoperative hypoxemia5-9.
The underlying pathophysiological mechanisms of impaired oxygenation during anesthesia include venous admixture increase (shunt) and ventilation to perfusion disturbances, that cause perfusion of lung areas with poor ventilation (low VA/Q units)1,3. In this concept, airway closure and formation of atelectasis can, in most cases, explain the oxygenation impairment.
Development of atelectasis during anesthesia and its implication on intraoperative hypoxemia has been originally proposed more than 50 years ago, by Bendixen et al in their classical study. The authors of this study concluded on the formation of atelectasis as a cause of impaired oxygenation10. Since then, atelectasis formation and its detrimental effects on intraoperative oxygenation have been confirmed in many clinical and experimental studies11-13. Atelectasis is seen in the majority of patients (90%) who are anesthetized, regardless of the ventilation mode or the anesthesia technique applied. Collapse of the lung tissue is associated with an increase in pulmonary shunt and it seems that there is a direct correlation between the size of the atelectatic lung parenchyma and the impairment of oxygenation13-19.
As far as the pathophysiological background is concerned it seems that the main underlying responsible mechanism of intraoperative atelectasis formation is compression20. Compression mechanisms include among others cranial shift of the diaphragm, alterations in the geometry of the thorax cavity and the kinetics of the diaphragm and blood volume displacement. Gas resorption behind collapsed airways and secondarily loss of surfactant may have an additional effect on the collapse of the lung tissue20,21.
Prevention and treatment of atelectasis have been investigated thoroughly by many research groups during the last years. Most of the studies have been conducted on patients with Acute Lung Injury or Acute Respiratory Distress Syndrome, who suffered severe disturbances of the lung mechanics and critical gas exchange impairment22,23.
Application of Positive End Expiratory Pressure (PEEP) and recruitment maneuvers are proposed as two of the most effective interventions to treat atelectasis24-27.
As far as PEEP is concerned, there are many parameters to be considered that might influence its effectiveness, such as level and duration of PEEP application and possible cardiovascular adverse events. It seems that sole PEEP application is not always adequate to treat atelectasis28.
Recruitment maneuvers are proposed as another promising intervention. However, the airway pressure level achieved determines the efficacy of this technique29,30. Indeed, we should always consider the unfavorable hemodynamic consequences31,32.
Despite all positive results described in the literature about application of the above mentioned methods, they have not yet been introduced in daily clinical anesthesia routine for treatment of atelectasis and improvement of arterial oxygenation30,33,34.
The aims of our study were to elucidate the percentage of intraoperative oxygenation impairment in patients with healthy lungs, undergoing elective surgery, and to investigate the efficacy and safety of different recruitment maneuvers.
Patients and methods
A total number of 430 patients scheduled for elective general surgery under general anesthesia were studied for impaired intraoperative oxygenation, which was defined as drop of PaO2/FiO2 ratio below 300. Out of these 430 patients, 150 actually developed oxygenation impairment during anesthesia and were enrolled in this study addressing the efficacy of recruitment maneuvers.
All 150 patients were free from preexisting respiratory disease and were submitted to elective surgery under general endotracheal anesthesia and mechanical ventilation. Exclusion criteria were: preexisting respiratory pathology based on medical history, physical findings and spirometry values (80% below predicted values), ASA physical status≥4 and the predicted or unexpected need for postoperative mechanical ventilation.
After Ethical Committee approval for this study, written informed consent was obtained during preanesthetic evaluation. Along with standard monitoring (electrocardiography, pulse oximetry and non-invasive blood pressure measurement) all patients had an arterial line inserted with topical anesthesia with lidocaine 2% for arterial sampling in room air prior to induction to anesthesia. All patients had continuous capnography and airway pressure measurements (Peak Inspiratory Pressure, Plateau Pressure, Mean Pressure) throughout anesthesia.After preoxygenation with face mask and oxygen flow at 10lt/min, anesthesia was induced with propofol 1.5-2.0mg/kg BW (Body Weight), lidocaine 1mg/kg BW, fentanyl 4mcg/kg BW and cisatracurium 0.15mg/kg BW. After endotracheal intubation, volume controlled mechanical ventilation was applied with Primus Anesthetic Workstation (Drager Medical). Ventilatory settings were the following: Tidal Volume = 8-10ml/kg BW, Respiratory Rate = 9-10/min targeting normocapnia, Fraction of Inspired Oxygen (FiO2) = 1.0 during induction and 0.5 immediately after intubation and Positive End Expiratory Pressure (PEEP) = 5cmH2O. Anesthesia was maintained with sevoflurane, fentanyl and cis-atracurium as needed.
Measured parameters of the total number of patients are depicted on Table 1.
Table 1. Measured parameters
| Hemodynamic parameters | Blood gas analysis | Respiratory parameters |
|---|---|---|
| Heart Rate (HR) | PaO2 | EndTidal CO2 |
| Systolic Arterial Pressure (SAP) | PaO2/FiO2 | Peak Inspiratory Pressure (PIP) |
| Diastolic Arterial Pressure (DAP) | SpO2 | Pplateau |
| Mean Arterial Pressure (MAP) | SaO2 | Pmean |
| Heart Rate (HR) | PaCO2 | Compliance |
Measurements were performed before intubation, 5 minutes after intubation and initiation of mechanical ventilation and every 30 minutes until the end of surgery. Furthermore, whenever a SpO2 reduction by 2% was observed, arterial blood gas sampling was performed in order to confirm the oxygenation derangement. Any reduction of the PaO2/FiO2 ratio below 300 was considered as impaired intraoperative oxygenation according to the study protocol and therefore the patient was enrolled in the study and was randomly allocated into one of the four study groups, as listed on Table 2.
Table 2. Study groups
| Groups | n | Intervention |
|---|---|---|
| A | 38 | Three hyperinflations of the lungs at an airway pressure of 40cmH2O for 10 sec each + increase in PEEP from 5 to 10cmH2O. |
| B | 38 | Increase in PEEP from 5 to 10cmH2O |
| C | 37 | Three hyperinflations of the lungs at an airway pressure of 40cmH2O for 10 sec each, without any PEEP alteration |
| D | 37 | No maneuver |
For the patients with oxygenation impairment, the parameters listed on Table 1 were measured at standard study phases, which are listed on Table 3.
Statistical analysis: The statistical program SPSS 19 was used for statistical analysis. All measured parameters are expressed as mean values plus standard deviation. The normality of data distribution was confirmed by Kolmogorof-Smirnof testing. One way ANOVA and x-square analysis were applied for the comparison of demographic data between groups. ANOVA for repeated measurements and Bonferroni test were applied for the comparison of measured parameters between groups and p<0.05 was considered statistically significant.
Table 3. Phases of measurements
| 0 | Oxygenation impairment |
|---|---|
| 1 | 5 min after intervention |
| 2 | 15 min after intervention |
| 3 | 30 min after intervention |
| 4 | 45 min after intervention |
| 5 | 60 min after intervention |
| 6 | End of surgery-pre extubation |
Results
In total, 430 patients scheduled for elective general surgery under general anesthesia and mechanical ventilation, were studied for oxygenation impairment. Surgical procedures performed included upper abdominal (n=137), lower abdominal (n=63), laparoscopic (n=40), extra peritoneal (n=80) and neck (n=110) procedures. Out of the 430 patients, 150 patients (38.4%), 74 male and 76 female, of mean age 62.2±13.4 developed oxygenation impairment during anesthesia and were randomly allocated into one of the four study groups.Demographic data and characteristics of these patients are presented in Table 4.
Table 4: Patients’ demographic data and co morbidities
| GROUP A | GROUP B | GROUP C | GROUP D | |
|---|---|---|---|---|
| n | 38 | 38 | 37 | 37 |
| Age | 57.3±15.4 | 64±12.7 | 61.4±10.4 | 65±13.9 |
| Gender M/F | 21/17 | 17/21 | 13/24 | 23/14 |
| Weight | 90.5 ± 26.4 | 82.3 ± 16.4 | 76.6 ± 13.8 | 81.2 ± 16.2 |
| BMI | 31.3±8.2 | 29.8±5.1 | 28.6±4.8 | 28.6±4.6 |
| ASA-PS, I/II/III | 2/27/9 | 0/18/20 | 1/21/15 | 0/16/21 |
| Smoking Yes/No | 13/25 | 11/27 | 13/24 | 15/22 |
| AH, Yes/No | 19/19 | 21/17 | 20/17 | 23/14 |
| CAD Yes/No | 9/29 | 13/25 | 9/28 | 10/27 |
| DM, Yes/No | 6/32 | 8/0 | 6/31 | 3/34 |
No statistically significant differences of the demographic data were observed between the groups.
Timing of oxygenation impairment occurrence was 33.7±26.2 minutes (CI: 29.5 – 37.9, Median: 30) after intubation and initiation of mechanical ventilation.
The descriptive statistics of PaO2 values of the four study groups at the various study phases are presented in Table 5.
Data are expressed as mean ± standard deviation.
Table 5. PaO2, Descriptive statistics
| 95% Confidence Interval Mean | |||||
|---|---|---|---|---|---|
| PHASES | GROUPS | MEAN | SD | Lower Bound | Upper Bound |
| 0 | A B C D | 123,2 124,3 126,1 128,9 | 18,6 22,9 23,2 14,9 | 117,1 116,9 118,3 123,9 | 129,3 131,9 133,8 133,9 |
| 1 | A B C D | 228,4 144,9 173,6 128,9 | 33,4 38,6 44,2 14,9 | 217,4 132,2 158,8 123,9 | 239,4 157,6 188,3 133,9 |
| 2 | A B C D | 209,4 157,8 145,2 123,5 | 33,4 42,2 39,9 10,9 | 198,4 144,1 131,9 119,9 | 220,4 171,7 158,6 127,2 |
| 3 | A B C D | 217,6 161,7 136,5 122,9 | 30,3 39,1 27,1 20,1 | 207,4 148,9 127,5 116,3 | 227,9 174,6 145,5 129,6 |
| 4 | A B C D | 213,4 172,4 125,4 116,1 | 27,4 36,1 26,4 15,9 | 202,9 159,8 116,1 110,6 | 223,8 184,9 134,8 121,6 |
| 5 | A B C D | 215,3 181,4 119,4 113,8 | 30,2 31,6 22,3 15,8 | 203,2 169,5 110,1 108,4 | 227,6 193,2 128,9 119,2 |
| 6 | A B C D | 218,8 190,9 121,7 114,1 | 28,3 41,6 18,6 15,1 | 209,5 177,3 115,5 109,1 | 228,1 204,6 128,1 119,1 |
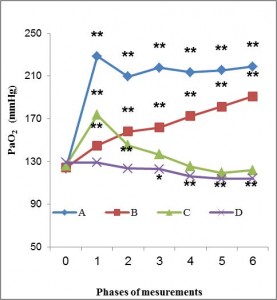
Figure 1. PaO2 alterations over time. Asterisks indicate statistical significance versus baseline *p<0,05, **p<0,001. In Groups A and B, PaO2 values differ statistically significant at all study phases compared to baseline, in Group C, PaO2 values differ statistically significant only at 5 and 15min (phases 1 and 2) compared to baseline, whereas in Group D, PaO2 values differ statistically significant only at 15, 30, 45 and 60min (phases 3-6) compared to baseline.
Descriptive statistics of PaO2/FiO2 ratio at the various study phases are depicted on Table 6.
Table 6. PaO2/FiO2 Ratio, Descriptive statistics
| 95% Confidence Interval Mean | |||||
|---|---|---|---|---|---|
| PHASES | GROUPS | MEAN | SD | Lower Bound | Upper Bound |
| 0 | A B C D | 246,4 248,7 252,1 257,8 | 37,3 45,8 46,5 29,8 | 234,1 233,6 236,6 2475 | 258,7 263,8 267,6 267,8 |
| 1 | A B C D | 456,9 289,8 347,2 257,8 | 66,9 77,3 88,5 29,8 | 434,9 264,4 317,7 247,9 | 478,9 315,2 376,7 267,8 |
| 2 | A B C D | 418,9 315,7 290,5 247,1 | 66,9 84,3 79,9 21,8 | 396,9 288,1 263,8 239,8 | 440,9 343,4 317,2 254,4 |
| 3 | A B C D | 435,3 323,5 273,1 245,9 | 60,6 78,2 54,1 40,1 | 414,8 297,8 255,1 232,5 | 455,8 349,2 291,1 259,3 |
| 4 | A B C D | 426,8 344,8 250,9 232,2 | 54,8 72,1 52,8 31,8 | 405,9 319,6 232,1 221,3 | 447,7 369,9 269,6 243,2 |
| 5 | A B C D | 430,7 362,8 238,9 227,6 | 60,4 63,3 44,7 31,6 | 406,3 339,1 220,1 216,7 | 455,2 386,5 257,8 238,5 |
| 6 | A B C D | 437,7 381,9 243,5 228,0 | 56,6 83,3 37,3 30,1 | 419,1 354,5 231,1 218,1 | 456,3 409,3 256,1 238,1 |

Figure 2. PaO2/FiO2 ratio alterations over time.
Asterisks indicate statistical significance versus baseline *p<0.05, **p
Asterisks indicate statistical significance versus baseline *p<0.05, **p<0.001.In Groups A and B, PaO2/FiO2 values present statistically significant difference at all study phases compared to baseline. In Group C, PaO2/FiO2 values present statistically significant difference only at phases 1 and 2 compared to baseline, whereas in Group D, PaO2/FiO2 values present statistically significant difference at phases 3-6 compared to baseline.
During the recruitment maneuvers in Groups A and C, a significant reduction of the systemic arterial pressure was observed, which was immediately restored at the end of the hyperinflations and was therefore not measured at phase 1, which was 5min after the maneuvers. None of the patients presented a significant hemodynamic impairment that would impose cessation of the recruitment strategy.
Hemodynamic parameters did not differ statistically significantly between the study groups, with the exception of diastolic arterial pressure (DAP), which was significantly higher in Groups A and C. However, this difference was of no clinical importance and the hemodynamic parameters remained in all patients within normal values throughout the study period.
Discussion
General anesthesia and mechanical ventilation are associated with impaired oxygenation even in patients without any lung dysfunction. The clinical impact of intraoperative hypoxemia is of great importance and represents a significant cause of perioperative morbidity and mortality. Prevention and efficacious treatment of intraoperative oxygenation derangement remains a continuous challenge for the anesthetist35, 36.
The purpose of this randomized clinical trial was the recognition of intraoperative oxygenation impairment among the general surgical population and the evaluation of three different recruitment strategies for the improvement of oxygenation.
Out of a total number of 430 patients, who underwent general surgery procedures under general anesthesia and controlled mechanical ventilation, 150 patients developed intraoperative oxygenation impairment and were included in this study.
According to the classical definition, hypoxemia is considered mild when SpO2<90% and severe when SpO2<85%.37 However, it is well known that these SpO2 values correspond to a wide range of PaO2 values, some of which reflect a clinically important impairment of gas exchange. Moreover, not all patients can tolerate the same low SpO2 values and many other factors are implicated and influence the occurrence of clinically important hypoxemia37.
Therefore, in order to achieve a more strict assessment of impaired oxygenation in our study, we decided on the PaO2/FiO2 ratio<300 to define oxygenation impairment. This ratio is also used for the Acute Lung Injury definition38-41.
Based on this criterion and according to the results of our study we reached the conclusion that almost 1 out of 3 patients with healthy lungs, undergoing elective surgery, will develop intraoperative oxygenation impairment. This percentage might be lower compared to some other studies in the literature42. This finding could be explained by the strict selection of patients with healthy lungs in our study and the standard application of PEEP in all patients immediately after initiation of mechanical ventilation, according to our protocol43.
Intraoperative oxygenation impairment occurred at approximately 30 minutes after intubation and initiation of controlled mechanical ventilation (33.7 ± 26.2, CI: 29.5 – 37.9, Median: 30).
According to the study protocol, patients who developed oxygenation impairment were randomly divided into 4 groups (Table 2). In Groups A, B and C three different recruitment strategies were applied and evaluated, whereas Group D was the control group. Measurements were performed at 6 different phases, whereas phase 0 was the baseline measurement (Table 3). Statistical analysis of data showed that PEEP increase from 5 to 10cmH2O in Group B led to a gradual but moderate improvement in oxygenation. The positive effects of PEEP in pulmonary function are well known and described in several studies in the literature. Tokics et al concluded that the application of 10cmH2O PEEP in a small number of patients contributed to reversal of the lung densities in the CT scan44. However, it did not seem to have any effect on oxygenation42. In another more recent study, Pelosi et al reported that PEEP application improves pulmonary function in obese patients but not in normal weighted ones45.
Standard application of 5 cmH2O of PEEP to all patients seems to have decreased the percentage of oxygenation impairment, compared to other studies42, but did not manage to completely prevent it, since 5cmH2O of PEEP are possibly not enough to keep the lung open and prevent atelectasis. On the other hand it is quite difficult in the intraoperative setting to determine optimum PEEP for each patient36,46. It must also be noted that in most literature studies PEEP application is compared to ZEEP (Zero End Expiratory Pressure), whereas in our study all patients received a standard minimum PEEP of 5cmH2O47,48.
Application of three hyperinflations in Group C without any PEEP modification led to an immediate improvement in oxygenation but the positive effect was of no significant duration. The oxygenation index presented a statistically significant difference only at phase 1 and 2 compared to baseline value.
These findings contrast with those described by Rothen et al, who observed that application of a vital capacity maneuver (inflation of the lungs up to 40cmH2O) is an effective technique to re-open atelectatic lung regions and to improve oxygenation in normal weighted patients30,33.
Application of three hyperinflations combined with an increase of PEEP from 5 to 10 cmH2O in Group A, led to a significant improvement in oxygenation. Moreover, this improvement remained statistically significant compared to baseline value at all different study phases and throughout the end of the study. These results come in accordance with other studies, which showed that recruitment maneuvers followed by PEEP reduced atelectasis and improved oxygenation, whereas recruitment maneuvers or PEEP application alone showed no significant results49-53.
Finally, in the control group (Group D), where no intervention was applied, the measured parameters of PaO2 and PaO2/FiO2 presented a gradual deterioration without reaching the level of significant tissue hypoxia.
Post Hoc Test analysis revealed that intraoperative oxygenation progressed in a completely different manner over time in the 4 study groups, presenting a statistical significant difference among them.
According to our results, the 3 hyperinflations of 10sec duration followed by an increase in PEEP seem to excel all other recruitment methods described in our study in improving oxygenation. This recruitment maneuver is based on basic principles described in several studies of patients with Acute Lung Injury or Acute Respiratory Distress Syndrome22,23,54,55.
The results of these studies encouraged us to evaluate and elucidate the effectiveness and safety of this recruitment strategy in an extended number of surgical patients with healthy lungs. Our results are similar to those described by Tusman et al, who investigated a small number of patients with healthy lungs and concluded that an ‘alveolar recruitment maneuver’ followed by an increase in PEEP seems to be a beneficial intervention to correct gas exchange abnormalities during anesthesia34.
These effects can be attributed to the fact that vital capacity hyperinflations contribute to the re-opening of the alveoli and application of higher PEEP preserves this action and prevents re-atelectasis.
However, one possible adverse event that should not be ignored whenever intrathoracic pressure is increased is cardiocirculatory compromise. According to our results, the hemodynamic compromise was transient, since it was observed only at the time of the hyperinflation delivery and was immediately reversed when intrathoracic pressure returned to normal. It is well known, that hemodynamic response to increased intrathoracic pressures is strongly correlated to the intravascular volume status56. Therefore, adequate fluid administration is an absolute prerequisite in these patients57. However, in patients with known severe cardiac compromise, hyperinflation might cause clinically important hemodynamic adverse events despite sufficient volume replacement. In these cases, we could consider to halve the time of hyper-inflation to maximize safety.
In conclusion, there should always be a high clinical suspicion and alertness for the recognition and treatment of intraoperative hypoxemia, even in adults with healthy lungs undergoing elective surgery58,59. Moreover, under the specific conditions of the present study, it is suggested that the combination of 3 vital capacity hyper-inflations of 10sec duration followed by a PEEP increase from 5 to 10cmH2O is an efficient, safe and easily applicable recruitment strategy to treat impaired intraoperative gas exchange.
REFERENCES
- Hedenstierna G, Edmark L. The effects of anesthesia and muscle paralysis on the respiratory system. Intensive Care Med 2005; 31: 1327-35.
- Ehrenfeld J, Funk L, Van Schalkwyk J, et al. The incidence of hypoxemia during surgery : evidence from two institutions. Can J Anaesth 2010 ; 57: 888–97.
- Pedersen T, Viby-Mogensen J, Ringsted C. Anaesthetic practice and postoperative pulmonary complications. Acta Anaesthesiol Scand 1992; 36: 812-8.
- Celli BR, Rodriguez KS, Snider GL. A controlled trial of intermittent positive pressure breathing, incentive spirometry, and deep breathing exercises in preventing pulmonary complications after abdominal surgery. Am Rev Respir Dis 1984; 130: 12-5.
- Magnusson L, Spahn DR. New concepts of atelectasis during general anaesthesia. Br J Anaesth 2003; 91:61-72.
- Navajas D, Farre R, Rotger MM, et al. Effect of body posture on respiratory impedance. J Appl Physiol 1988; 64:194-9.
- Bindslev L, Hedenstierna G, Santesson J, et al. Ventilation-perfusion distribution during inhalation anaesthesia. Effect of spontaneous breathing, mechanical ventilation and positive end-expiratory pressure. Acta Anaesthesiol Scand 1981; 25: 360-71.
- Pelosi P, Croci M, Ravagnan I, et al. The effects of body mass on lung volumes, respiratory mechanics, and gas exchange during general anesthesia. Anesth Analg 1998; 87:654-60.
- Charuluxananan S, Suraseranivongse S, Punjasawadwong Y, et al. Risk factors of intraoperative oxygen desaturation: a case–control study of 152 314 anesthetics. J Med Assoc Thai 2007; 90: 2359-65.
- Bendixen H, Hedley-White J, CHir B, Laver MB. Impaired oxygenation in surgical patients during general anesthesia with controlled ventilation. N Engl J Med 1963; 269: 991-6.
- Gunnarsson L, Tokics L, Gustavsson H, et al. Influence of age on atelectasis formation and gas exchange impairment during general anaesthesia. Br J Anaesth 1991; 66: 423-32.
- Hedenstierna G, Lundquist H, Lundh B, et al. Pulmonary densities during anaesthesia. An experimental study on lung histology and gas exchange. Eur Respir J 1989; 2: 528-35.
- Strandberg Å, Tokics L, Brismar B, et al. Atelectasis during anaesthesia and in the postoperative period. Acta Anaesthesiol Scand 1986; 30: 154-8.
- Hedenstierna G, Tokics L, Strandberg A, et al. Correlation of gas exchange impairment to development of atelectasis during anaesthesia and muscle paralysis. Acta Anaesthesiol Scand 1986; 30:183–91.
- Hedenstierna G. Gas exchange during anaesthesia. Br J Anaesth 1990; 64: 507–14.
- Brismar B, Hedenstierna G, Lundquist H, et al. Pulmonary densities during anesthesia with muscular relaxation-a proposal of atelectasis. Anesthesiology 1985; 62: 422-8.
- Prys-Roberts, Nunn JF, Dobson RH, et al. Radiologically undetectable pulmonary collapse in the supine position. Lancet 1967; II: 399-401.
- Tusman G , Böhm S. Prevention and reversal of lung collapse during the intra-operative period. Best Practice & Research Clinical Anaesthesiology 2010; 24: 183 -97.
- Magnusson L, Spahn R. New concepts of atelectasis during general anaesthesia. BJA 2003 ; 92:61-72.
- Hedenstierna G, Rothen HU. Atelectasis formation during anesthesia: causes and measures to prevent it. J Clin Monit 2000; 16: 329-35.
- Varga M, Brunetti L Pelosi P. Protective mechanical ventilation during general anaesthesia. Trends in Anaesthesia and Critical Care 2013; 3: 77 – 81.
- Grasso S, Mascia L, Del Turco M, et al. Effects of Recruiting Maneuvers in Patients with Acute Respiratory Distress Syndrome Ventilated with Protective Ventilatory Strategy. Anesthesiology 2002; 96: 795-802.
- Patroniti N, Foti G, Cortinovis B, et al. Sigh Improves Gas Exchange and Lung Volume in Patients with Acute Respiratory Distress Syndrome Undergoing Pressure Support Ventilation. Anesthesiology 2002; 96: 788-94.
- Algaba A, Nin N. Alveolar recruitment maneuvers in respiratory distress syndrome. Med Intensiva. 2013;37:355-62.
- Ferrando C, Carbonell J, Gutierrez A, et al. Mechanical ventilation in the operating room: Adjusting VT, PEEP, and FiO2. Trends in Anaesthesia and Critical Care 2012; 2: 269 -73.
- Tusman G, Bohm S, Suarez-Sipmann F. Alveolar recruitment during mechanical ventilation. Where are we in 2013? Trends in Anaesthesia and Critical Care 2013; 3 : 238 -45.
- Tusman G, Belda J. Treatment of anesthesia-induced lung collapse with lung recruitment maneuvers Current Anaesthesia & Critical Care 2010: 21; 244-9.
- Hedenstierna G. Alveolar collapse and closure of airways: regular effects of anaesthesia. Clin Physiol & Func Im 2003; 23: 123-9.
- Rothen HU, Sporre B, Engberg G, et al. Re-expansion of atelectasis during general anaesthesia: a computed tomography study. Br J Anaesth 1993; 71: 788-95.
- Rothen HU, Neumann P, Berglund JE, et al. Dynamics of re-expansion of atelectasis during general anesthesia. Br J Anaesth 1999; 82: 551-6.
- Nielsen J, Nilsson M, Fredén F, et al. Central hemodynamics during lung recruitment maneuvers at hypovolemia, normovolemia and hypervolemia. A study by echocardiography and continuous pulmonary artery flow measurements in lung-injured pigs. Intensive Care Med 2006; 32: 585-94.
- Nielsen J, Østergaard M, Kjaergaard J, et al. Lung recruitment maneuver depresses central hemodynamics in patients following cardiac surgery. Intensive Care Med 2005; 31: 1189-94.
- Rothen HU, Sporre B, Engberg G, et al. Reexpansion of atelectasis during general anaesthesia may have a prolonged effect. Acta Anaesthesiol Scand 1995; 39: 118-25.
- Tusman G, Böhm S, Vazquez de Anda GF, et al. Alveolar recruitment strategy’ improves arterial oxygenation during general anaesthesia. Br J Anaesth 1999; 82: 8-13.
- Warner D. Preventing postoperative pulmonary complications: the role of the anesthesiologist. Anesthesiology 2000; 92:1467-72.
- Wirth S, Baur M, Spaeth B, et al. Intraoperative positive end-expiratory pressure evaluation using the intratidal compliance-volume profile. BJA 2015; 114: 483-90.
- Tusman G, Böhm S. Prevention and reversal of lung collapse during the intra-operative period. Best Practice & Research Clinical Anaesthesiology 2010; 24: 183 -97.
- Chen L, Marshall The diagnosis and management of perioperative hypoxemia. ASA Refresher Courses in Anesthesiology 1998; 16: 41-56.
- Bernard G, Artigas A, Brigham K, et al. The American–European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149: 818–24.
- Costa Ε, Amato Μ. The new definition for acute lung injury and acute respiratory distress syndrome: is there room for improvement. Curr Opin Crit Care 2013, 19:16–23.
- Raghavendran K, Napolitano L. Definition of ALI/ARDS. Crit Care Clin 2011; 27: 429 – 37.
- Moller J, Johannessen N, Berg H, Espersen K, Larsen L. Hypoxaemia during anaesthesia-an observer study. Br J Anaesth 1991; 66: 437-44.
- Neumann P, Rothen H, Berglund J, et al. Positive end-expiratory pressure prevents atelectasis during general anaesthesia even in the presence of a high inspired oxygen concentration. Acta Anaesthesiol Scand 1999; 43: 295–301.
- Tockics L, Hedenstierna G, Stranberg A, et al. Lung collapse and gas exchange during general anesthesia: Effects of spontaneous breathing, muscle paralysis, and positive end-expiratory pressure. Anesthesiology 1987; 66: 157-67
- Pelosi P, Ravagnan I, Giurati G, et al. Positive end-expiratory pressure improves respiratory function in obese but not in normal subjects during anesthesia and paralysis. Anesthesiology 1999; 91: 1221-23.
- Wirth S, Baur M, Spaeth B, et al. Intraoperative positive end-expiratory pressure evaluation using the intratidal compliance-volume profile. BJA 2015; 114 : 483-90.
- Wetterslev J, Hansen E, Roikjaer O, et al. Optimizing perioperative compliance with PEEP during upper abdominal surgery: effects on perioperative oxygenation and complications in patients without preoperative cardiopulmonary dysfunction. Eur J Anaesthesiol 2001;18: 358 – 65.
- Meininger D, Byhahn C, Mierdl S, et al. Positive endexpiratory pressure improves arterial oxygenation during prolonged pneumoperitoneum. Acta Anaesthesiol Scand 2005;49:778 – 83.
- Reinius H, Jonsson L, Gustafsson S, et al. Prevention of Atelectasis in Morbidly Obese Patients during General Anesthesia and Paralysis. Anesthesiology 2009; 111:979–87.
- Chalhoub V, Yazigi A, Sleilaty G, et al. Effect of vital capacity manoeuvres on arterial oxygenation in morbidly obese patients undergoing open bariatric surgery. Eur j Anaesthesiol 2007 ; 24: 283-8.
- Futier E, Constantin J, Pelosi P, et al. Intraoperative recruitment maneuver reverses detrimental pneumoperitoneum-induced respiratory effects in healthy weight and obese patients undergoing laparoscopy. Anesthesiology 2010; 113:1310 -9.
- Talab H, Zabani I, Abdelrahman H, et al. Intraoperative ventilatory strategies for prevention of pulmonary atelectasis in obese patients undergoing laparoscopic bariatric surgery. Anesth Analg 2009 ; 109: 1511-6.
- Whalen F, Gajic O, Thompson G, et al. The effects of the alveolar recruitment maneuver and positive end-expiratory pressure on arterial oxygenation during laparoscopic bariatric surgery. Anesth Analg 2006; 102: 298-305.
- Open up the lung and keep the lungs open. Intensive Care Medicine 1992; 18: 319 -321.
- Fan E, Wilcox M, Brower R, et al. Recruitment maneuvers for acute lung injury: a systematic review. Am J Respir Crit Care Med. 2008; 178:1156-63.
- Luecke T, Roth H, Hermann P, et al. Assessment of cardiac preload and left ventricular function under increasing levels of positive end-expiratory pressure. Intensive Care Med 2004; 30: 119-26.
- Fougéres E, Teboul JL, Richard C, et al. Hemodynamic impact of a positive end-expiratory pressure setting in acute respiratory distress syndrome: importance of the volume status. Crit Care Med 2010; 38 : 802-7.
- Talley H, Bentz N, Georgievski J, et al. Anesthesia Providers’ Knowledge and Use of Alveolar Recruitment Maneuvers . J Anesth Clin Res 2012; 3: 1-5.
- Hedenstierna G, Edmark E. Mechanisms of atelectasis in the perioperative period. Best Pract Res Clin Anaesthesiol. 2010; 24: 157-169.
Author Disclosures:
Authors Veroniki F., Ourailoglou V., Fyntanidou B., Tsotsolis N., Kotso E., Kyparissa M., Grosomanidis V. have no conflicts of interest or financial ties to disclose.
Corresponding author:
Fotini Veroniki
Anesthesiology Department, “O Agios Dimitrios” General Hospital Thessaloniki
Elenis Zografou 2
54634 Thessaloniki, Greece
Tel: +302313322181, +306932666101
E-mail:









