Authors: Aslanidis Th1 MD, Kontogounis G2 MD
1Mobile Intensive Care Unit, National Center of Emergency Care, Thessaloniki Dep., Greece
2Neurologist, Kozani, Greece
ABSTRACT
Clinical evaluation of pupils is considered as an essential part of neurological examination. The pupillary response to light is controlled by the autonomic nervous system. Numerous factors affect pupils dynamics, like e.g. luminance, visual field area, pain, drug administration, age, the functional integrity of anatomical structures involved, e.t.c. Moreover, pupillometry card method and examination of pupil reaction with the use of a penlight is subjective to a lot of bias. Portable infrared pupillometry allows a more objective and detail evaluation of pupil’s dynamics. That’s why it has already found applications in various clinical areas, like e.g. neurology, psychology, ophthalmology, endocrinology, anesthesia, pain management, intensive care, emergency medicine. This review focuses on physiology of pupil’s dynamics and on applications of infrared pupillometry in perioperative setting.
INTRODUCTION
Archimedes was the first to report observations about the size and the symmetry of eye’s pupil and Galileo is believed to be the first who attempted to measure its diameter1. Pupillary reflex dilation was originally described by Budge in 1852 as a sympathetic spinal reflex that dilated the pupil after noxious stimulation2. In 1885 Bellerminov published static records of pupil’s shape under side light3. In 1942 Lowenstein presented dynamic records of pupil’s changes under ultraviolet and infrared light emission. Later, Matsunaga (1973) and Jones (1983) developed recording systems of pupil’s movement; but the latter were cumbersome and thus, impossible to be used in clinical setting4-6 .
Despite the difficulties of an objective bedside measurement, manual (performed using a penlight or ophthalmoscope) pupil evaluation remains an essential part of neurological clinical examination for over a century now1.
The recent emergence of new technologies offered us a new perspective as they introduced us to the era of digital pupil evaluation. The present article focuses on digital portable pupillometry and it reviews the possible applications of this technology in perioperative field.
Anatomical and physiological base of pupil’s dynamic
The pupils are dilated in excitement, in the dark, and with mydriatic agents like atropine; conversely they are constricted in neonates, during sleep, with ocular convergence as part of the near reflex, with use of the miotic agents like acetylcholine and pilocarpine, and also they are constricted with light directed toward the eye. In fact, the most impart factor for the pupillary size is the amount of ambient light.
The size of the pupil of the eye is determined by the balance between the tone of two muscles, the constrictor (sphincter, the circular muscle) and the dilator (the radial muscle). The light reflex (PLR)is the simultaneous and equal constriction of the pupilin response to illumination of one (direct) or the other eye (indirect or consensual). The reflex is multisynaptic with pulse speed 160 m/sec and triggering time about 200msec. The afferent pupillary pathway is controlled by the sympathetic autonomous nerve system (ANS) and originates in the retina. The axons of retinal ganglion cells pass into the optic nerve, partially (only the 2nd, 3rd, 5th and 6th laminae) decussate in the chiasm and continue (optic tract) to the midbrain (lateral geniculate body), while some pupillary fibers bypass the ciliospinal center and continue cephalad to the pretectal nuclei at the level of the superior colliculus (thalamus). Optical cortex include Brodmann areas 17,18 and 19 (occipital lobe) and inter-connection with frontal lobe (Brodmann area 8) and several nuclei of cranial nerves (oculomotor, trochlear and abducens), (Fig. 1).
- Figure 1.a The neural efferent optical path-way.
- Figure 1.b. The neural efferent optical path-way.(From http://en.wikipedia.org/wiki/Pupillary_light_reflex)
Considering the efferent arc of the light reflex, the visceral nuclei of the oculomotor nucleus, the nuclei that are believed to contain cell bodies of preganglionic, parasympathetic neurons that project to the ciliary ganglion and synapse with neurons that eventually produce both pupillary constriction and accommodation, are located in the dorsal midbrain. These consist of the Edinger-Westphal nuclei (also called the dorsal visceral cell columns), the anterior median nuclei, and the nucleus of Perlia. Preganglionic parasympathetic fibers run in the oculomotor nerve as it leaves the brain stem. The fibers pass downward to lie inferiorly in the inferior division of the third nerve as it enters the orbit. These fibers synapse in the ciliary ganglion and give rise to postganglionic parasympathetic myelinated short ciliary nerves, about 3%-5% of which are pupillomotor. The rest are designated for the ciliary muscle and are concerned with the near reflex, (Fig. 2a and 2b).
 CCN, central caudal nucleus; CN III, oculomotor nerve; IO, inferior oblique; IR, inferior rectus; LP, levatorpalpebraesuperioris; MR, medial rectus; MRa, medial rectus subnucleus a; MRb, medial rectus subnucleus b; MRc, medial rectus subnucleusc; SR, superior rectus. (From Ksiazek S, Slamovits TL, Rosen CE, et al. Fascicular arrangement in oculomotor paresis. Am J Ophthalmol 1994;118: 97–103). |
Figure 2.a Schematic drawing of position of fibers in fascicle of human oculomotor nerve. Fibers destined for pupillary sphincter (P) oc-cupy a rostral and medial position in the fasci-cle.
|
A, dorsal and B, ventral side of brain stem. P, pons; M, medulla; EW, Edinger-Westphal nucleus; IIIn, somatic portion of third nerve nucleus; III, third nerve;ID, inferior division of third nerve; SD, superior division of third nerve; NCilV, nasociliary branch of fifth nerve; CG, ciliary ganglion; Sym, sympathetic route; m, medial; l, lateral. (From Kerr FWL, Hollowell OW. Location of pupillomotor and accommodation fibres in the oculomotor nerve: experimental observations on paralytic mydriasis. J NeurolNeurosurg Psychiatry1964; 27:473.).

Figure 2.b Course of preganglionic autonomic nerve fibers from the brain stem to the ciliary gan-glion. A sagittal reconstruction of the brain stem with the course of the oculomotor nerve is shown at top. The corresponding locations of the preganglionic autonomic fibers for pu-pilloconstriction and accommodation within the right (R) and left (L) oculomotor nerves are shown in black in coronal sections through slices at 1 (emergence from the brain stem), 2 (midpoint in the subarachnoid space), 3 (at the point where the third nerve enters the dura), and 4 (in the anterior cavernous sinus where the fibers have entered the anatomic inferior division of the third nerve). The autonomic fibers are located superiorly as the oculomotor nerve exits the brain stem and then come to lie more medially as the oculomotor nerve passes toward the orbit.
Factors that determine the pupillary characteristics. The difficulty of objective measurement.
In a young observer, pupil diameter may vary between 2-8 mm (i.e. a 16-fold change) with variations in light level. In the majority of cases both pupils are equal. However, about 20% of individuals have noticeably different diameters in their two pupils; a phenomenon known as physiologic (also simple or essential)anisocoria.
Pupil size is affected by diverse intrinsic and extrinsic factors (table 1). The variations in pupil diameter trigger changes in retinal stimulation, relevant to retinal function: changes in retinal illuminance, the ratio of rod/cone stimulation, spectral sensitivity and spatial resolution7 (Fig. 3).
Figure 3. Several determinants and effects of pupil size.
- a. Luminance
- b. Light wavelength (red 470nm & blue 660 nm)
- c. Distance form light source (Based on Sacek, Vladimir. “13.9. Eye Spectral Response.” Notes on AMATEUR TELESCOPE OPTICS. Vladimir Sacek, 14 July 2006. http://www.telescope-optics.net/index.htm, Accessed Web. 18 Aug. 2014.)
Table 1.Factors affecting pupil size.
| Stimulus variables
-light level -spectral composition -spatial configuration: field size, spatial structure of the field -monocular/binocular view -accommodative state -non visual stimuli : pain, noise |
| Observer variables
-individual differences -age -day-to-day within observer variance -biomechanical factors: respirations, heart beat -cognitive factors: arousal, attention, fright, workload, hedonistic content |
The complexity of the aforementioned anatomical and physiological pathways should be kept in mind when assessing pupil size and reactions. For example, in case of a lesion of the optic nerve anterior to the chiasm, there will be a combination of vision loss, color vision loss (dyschromatospia) and visual field defect. If the light is directed to the affected eye, there will be less retinomesencephalic input and parasympathetic tone will decline, i.e. less relative miosis in both eyes. The contrary will happen if the light is directed to the unaffected eye.
Fast or slow eye movements how are control by the oculomotor system which has the goal to keep the object of regard on the fovea, nystagmus, saccadic intrusions and oscillations can also interfere with pupils’ assessment. Yet, further analysis of these mechanisms is beyond the scope of this article.
LeGrand formulated an equation estimating the average pupil diameter from stimulus luminance and taking account of the loss of photopic efficacy of light entering the eccentric portions of the pupil (the Stiles-Crawford effect): d= 5-3 tanh (0.4logL), whered is pupil diameter in mm and L is luminance in cd/m2 7-8.
Moreover, in conditions of permanent lighting and eye fixation, a dynamic equilibrium of pupil size-for which the sympathetic and parasympathetic activity modulated by central nervous system is responsible-is noticed; the phenomenon of spontaneous pupillary fluctuation9 (Fig. 4).
Spectral determinations of tonic pupil size include also field size and light level. For fields >5o, pupil size is predominately determined by the scotopic luminous efficiency function V’ (λ) for which the rods are active. For fields <2o, pupil size is predominately determined by photopic luminous efficiency function V (λ) 10.
Literature data also indicate that pupil diameter is smaller with binocular view, but report differ on the magnitude of the phenomenon (x2 to x10)7.
Finally, pupil diameter is found to become small in old age (involutionalmiosis). In fact, its diameter decreases by 0.043 mm/yr at 9cd/m2 and by 0.015 mm/yr at 4400cd/m2 11(Fig. 5).
Combining one or more of the aforementioned factors, a number of formulas have been proposed to describe the light-adapted pupil size. One of the most recent is the Watson-Yellot equation: Du (L, a, y, yo, e) = DSD (F, 1) + (y-yo) [0.02132 – 0.009562 DSD (F, 1)where L is kuminance, a is field area in deg2, y is age in years, y0 is the reference age, and e is the number of eyes (one or two). The reference formula for the above mentioned equation is the Stanley-Daves equation, thus the reference age should be the mean age of the population of observers used by Stanley-Davies; and F is effective corneal flux density that describes the quantity that effectively controls pupil diameter, equal to the product of luminance area and the monocular effect (F=LaM(e))12. Further analysis of the concept behind the development of these equations is beyond the scope of this article.
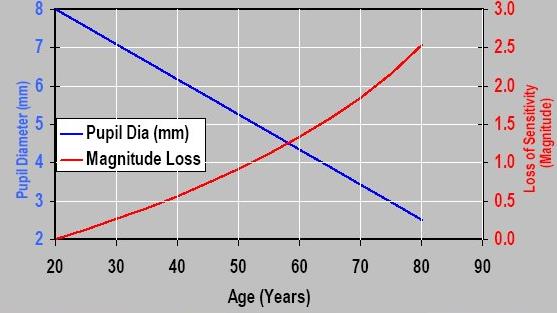
Figure 5. Reduction of eye’s pupil.Loss of eye sensitivity (fue to reduction in exit pupil) with age.
How then can we know pupil size?
When examining the PLR, attention should be directed to any difference in size (anisocoria), shape (dyscoria), or position (corectopia). Strategies for measurement include direct comparison with a scale, entopic method (visualize a shadow of own pupil from near point source) and infrared pupillometry. The first is easy but subjective and prone to error. A pupillary inequality of 0.2 mm can be detected clinically, and anisocoria of 0.4 mm is readily visible. Pupil light reactivity is graded as “brisk”,”sluggish” and “nonreactive”. The reflex is more difficult to detect with a wide pupil (e.g. a 0.2 mm reflex in a 5 mm diameter pupil is only a 4% reflex, whereas the same reflex in a 2 mm diameter pupil is a 10% reflex) and in a dark iris (as the ambientlight must be high for the iris to be seen, which reduces the step increase induced by the penlight).If the pupillary light reflex amplitude is less than 0.3 mm and the maximum constriction velocity is less than 1 mm/s, the reflex is unable to be detected using a penligh13. The rest of the pupil characteristics (velocity of response, location, presence of pathologic movements) estimation is based on observer experience (table 2). Entopic method, though it uses a flashlight and a pinhole and it’s easy to perform, it interferes with steady state pupil size.
Table 2. Types of pupils’ disturbances
| Name | Description | Disturbance |
|---|---|---|
| Argyll–Robertson | Small irregular shaped pupils that react (constrict) to accommodation but not to direct light | Neurosyphillis (tabesdorsales), Adie’s syndrome, Lyme’s disease |
| Marcus-Gunn (Swinging flashlight sign) | Normal bilateral light response when the light is shown into the intact eye; but when light is shown into the diseased eye it responds with dilation. | Lesions/atrophy of the retina or optic nerve. |
| Adie (TonicPupil) | Unilateral dilated pupil that reacts slowly to light after prolonged stimulation with slow accommodation. | Postganglionic denervation of the parasympathetic papillary innervation. Cause unknown; may be seen post viral infection.(Idiopathic internal ophthalmoplegia) |
| Hutchinson | Mydriatic pupil with no reaction to light. | Intracranial hypertention |
| Hippus | Bilateral synchronous and spontaneous rhythmic constriction and dilatation, with large (>1mm) amplitude. | Barbiturate, Aconita poisoning, metabolic encephalophathy, epilepsy, can be observed in healthy persons |
| Miotic Pupils (Miosis, or Small pupils < 2mm, Pin point pupils) | Bilateral or unilateral small pupils. | Certain drugs such as acethlcholine chloride, carbachol, cemecarium bromide, echothiphate iodide, isoflurophate, physostigmine,pilocarpine, and narcotics. Also due to pontine hemorrhage or infarct, direct orbital injury to the eye with destruction of the sympathetic innervation and sleep |
| Amaurotic Pupil (Blindeye) | No direct light response, no consensual response in the blind eye | Disease of the retina or optic nerve |
| Mydriatic Pupil (Large Pupils > 6mm in size) | Bilateral or unilateral large pupils. | Certain drugs: hallucinogens,antihistaminesglutethiamide, anticholinergics, and dopamine. Direct eye trauma to nerve endings of the iris sphincter muscle |
Considering the aforementioned, it is not surprising that there are reports which describe inter-observer variability up to 34% 14-18.
Why insisting in pupil’s examination?
First of all, because we can assess the functionality of the optical structures; but most importantly, because the pupil’s dynamics reflect the functional balance between sympathetic and parasympathetic ANS. In fact, in absence of injuries of the peripheral structures, pupillometry is an indirect estimation of functional status of ANS (especially at midbrain level). In combination with other methods of ANS examination (e.g. electrodermal activity) it has found various applications19: for the evaluation of injury of parasympathetic ANS in alcoholics20,in pain study21, in patients with diabetes mellitus22, in cases of severe myasthenia23, Down syndrome24-25, Alzheimer’s disease24, depression22, mental retardation26, in Parkinson disease27,32, in cases of drug overdoses28-29or patients with collagen disease30-32.
In addition, pupil examination is related with differential diagnosis of coma33, and it is used as part of prognostic systems of survival after trauma brain injury34-36 and as vital index in management therapeutic strategy in cases of both early and late brain trauma37-40. Today, it is included in all life support protocols and it is considered as an additional part of Coma Glasgow scale41-43.
The problem
The previous sections highlight the necessity of an objective method of pupil examination. In fact, subjective pupil evaluation may compromise large multicenter trials’ results, like e.g. DECRA trial for decompresisvecraniectomy in traumatic brain injury43-44.
Infrared pupillometry
Infrared pupillometry is generally classified in photographic, which does not use online information and requires frame-by-frame measurement, photoelastic (non-imaging), which requires calibration for each observer and video camera and computer –assisted which seems to be the most accurate. Unfortunately, till recently, these systems were considered cumbersome and expensive. Today, the emergence and availability of portable pupillometers seems to offer a solution to the problem (image 1). The basic concept: automatic evaluation of pupil response to light. The duration of emission is 100msec, while the rest of the test parameters are manually selected.
Image 1. Different models of portable pupillometers

(for left to right):a. VIP 200 (NeuroOptics, Irvine, CA,USA), b: NPi-100 (NeurOptics, Irvine,CA,USA), c: EYE check (MCJ Inc.,USA) and d: Colvard Pupillometer (Optical Inc, USA), AlgiScan (iDMed, Marseille, France).
With infrared pupillometry intra-examiner variability can drop down to 1%45-47. Apart from that, the development of quantitative scale of pupil reaction (NPi-Neurological Pupil Index) allows a more rigorous and classification of pupil response (image 2).
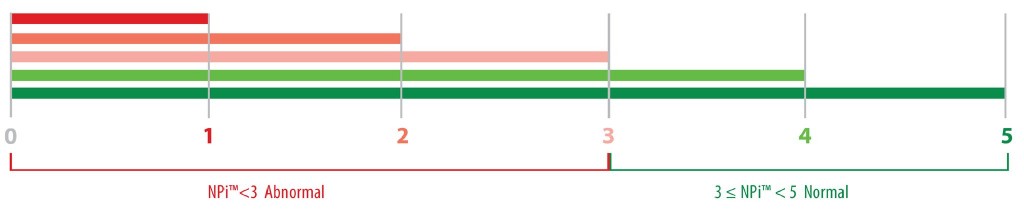
Image 2. The NPi index.Avaluebetween 3 and 5 means normal response while a value<3 suggest pathological response47.
Others parameters can be recorded to create a more complete picture: the resting (maximum) and the minimum diameter, the percentage change(%Ch=maximum-minimum/maximum), the latency (time difference between of retinal light stimulation and onset of pupillary constriction), the average and maximum constriction velocity and the average dilatation velocity (image 3)47. Other studies report more parameters, like e.g. maximum re-dilatation velocity, the interval till the 75% of the re-dilatation (t75%), and the relative reflex amplitude48. Results from new studies are expected.
These measurements allow not only the evaluation of pupil’s diameter but also the neurophysiology of its reaction. Thus, e.g. latency reflects the functionality of the nerve impulse transfer; the maximum constriction velocity reflects the parasympathetic ANS activation intense; the average dilation velocity reflects the sympathetic activation, e.t.c.
In anesthesiology, pupil reactions had been used as intraoperative algesimeter and as part of closed-loop monitor system of general anesthesia, along with other methods (entropy, bispectral index, etc). In the first case, a battery-operated device automatically measures, by means of an infrared camera, pupil diameter. This is measured in response to noxious stimulation produced by a tetanus at a frequency of 100 Hz for 5 seconds. Its intensity varies from 10 to 60 mA. The recording is performed for a period of 13 seconds (3 seconds prior to stimulation, stimulation for 5 seconds, 5 seconds after the stimulation). An interval of 3 minutes is allowed between the two measurements (right and left), which is significantly higher than the time necessary to return to normal (about 20seconds).
A new index, the pupillary pain index (PPI) ranges from 0 to 10 and is a composite measure of the pupillary diameter reflex to the tetanus stimuli delivered by the AlgiScan device. Smaller scores indicate increased analgesia50.
The overall use of these devices is easy and can be employed by both medical and paramedical personnel.
Limitations of the method include the relative high cost, the need for an open eye during the examination, ophthalmic diseases or operations and the lack of global collective experience and database.
Applications in perioperative setting
Among the first application were the conduction of studies for the drugs effect on pupil light reflex (Image 4), creating databases which can be used for noninvasive detection of drugs, even those administered epiduraly28, 49, 50.
Pupillometry seems more relevant than parasympathetic component of heart rate variability to assess analgesia during general anesthesia50. It had been with promising results used both for general and local anesthesia, both in adults and children51-54. Changes in PLR brought about by a uterine contraction may be used as a tool to assess analgesia in noncommunicating obstetrical patients as well55. Results from new studies (i.g. ALGISCAN Trial) both for intraoperative condition and postanesthesia setting are expected56-57.
Pupillometry has also been used for prediction outcome after cardiac arrests: it seems that early detection of PLR is associated with better outcome; moreover, PLR and has comparable prognostic accuracy than electroencephalogram (EEG) and somato-sensory evoked potentials (SSEP) .in predicting outcome of post-anoxic coma, irrespective of temperature and sedation58-63. Also in post-resuscitation non-brain dead critically ill patients with ‘absent’ pupillary reflexes, the reflex has been demonstrated using a portable infrared pupillometer58.
Likewise, there is growing literature about cases with “reversible fixed pupils” as it is realized that some of these conditions might be associated with ‘clinically undetectable’ rather than ‘absent’ pupillary light reflexes64. Other phenomena like, e.g. pupillary hippus have been associated with increased hospital mortality65 and with nonconvulsive status epilepticus in critically ill patients65, 66.
In neurosurgical critical care patients, there are several reports claiming that pupillometry can detect an early increase in intracranial pressure (ICP). A rise of ICP above the level of 20mmHg decrease the constriction velocity of ipsilateral to the injury pupil in values < 0.6 mm/sec (normal range 1.48 ± 0.33 mm/sec), while changes in NPi can be detected up to 15.9 hours before the ICP increase1,64,67-69.
Apart from that, PLR measurement has been used with good results as analgesia index in critically ill70-73.
Conlcusion–future perspectives
Infrared pupillometry complete an armamentarium of monitor tools for central neural system like transcranial Doppler, brain tissue monitoring, microdialysis, several specific (e.g. S-100 protein) biomarkers and other imaging examination (CT,MRI,fMRI,SPECT).
New studies are already investigating these fields74. Hence, a new era of neuromonitoring has begun, and it bring along great changes in future perception of conditions like anesthesia, sleep, coma, locked-in syndrome and brain mapping.
References
- Fountas KN, Kapsalaki EZ, MachinisTG, et al. Clinical implications of quantitative infrared pupillometry in neurosurgical patients. Neurocrit. Care 2005; 5: 55-60.
- Budge J. Uber den Einflus des nerven systems suf die Bewegung des Iris.ArchPhiolHeilk 1852; 2:773–9.
- Bellarminov L,Anwendung der graphischenMethodezurUntersuchungderPupillen-Bewegung.Pflugers Arch.Ges. Phusiol. 1885; 37:7.
- Lowenstein O, Lowenfeld E. Electronic PupillographyElectronic Pupillography, M. A. Arch. Ophthal.,1958; 59: 352-363
- Matsunaga K. A new Binocular Electronic Scanning Pupillometer, Physiologia 1973; 16: 115-120.
- Jones DP, Smith RA. A New Solid State Dynamic Pupillometer Using A Self-Scanning Photodiode Array. J Phys E. 1983; 16: 1169-72.
- Pokorny J, Smith VC. How much light reach the retina? In Color Vision Deficiencies XIII, Cavonious CR(ed.), Klower Acad. publishers, Dodreht, UK, 1997:pp. 491-511.
- LeGrand Y. Light, Color, Vision.2ndChapman& Hall, London 1968.pp.106.
- Nowa W, Hachol A, Kasprzak H. Time-frequency analysis of spontaneous fluctuation of the pupil size of the human eye. OpticaApplicata 2008; 43:469-80.
- Berman SM, Fein G, Jewett J, et al. Spectral determinants of steady-state pupil size with full field of view. J. Illum. Soc. 1992; 21: 12-33.
- Winn B, Whitaker D, Elliott DB, et al. Factors affecting light-adapted pupil size in normal human subjects. Invest. Ophthal. Vis. Sci. 1994; 35: 1132-7.
- Watson, AB, Yellott, JI. A unified formula for light-adapted pupil size. Journal of Vision, 2012; 12:1–16.
- Larson MD, Muhiudeen I. Pupillometric analysis of the ‘absent light reflex’. Arch Neurol 1995; 52:369-72.
- Clark A, Clarke TN, Gregson B, Hooker PN, Chambers IR.Variability in pupil size estimation. Emerg Med J. 2006; 23: 440-1.
- Litvan I, Saposnik G, Mauriño J, et al. Pupillary diameter assessment: need for a graded scale. 2000; 54:530-1.
- Wilson SF, Amling JK, Floyd SD, et al. Determining interrater reliability of nurses’ assessments of pupillary size and reaction. J NeurosciNurs 1988; 20:189-92.
- Witting MD. Validity of simple measurement to diagnose pupillary dilation. Am J Emerg Med. 2005; 23:155-8.
- Pop M, Payette Y, Santoriello E.Comparison of the pupil card and pupillometer in measuring pupilsize. J CataractRefractSurg.2002 ;28 :283-8.
- Bremner F. Pupil evaluation as a test for autonomic disorders.ClinAuton Res. 2009; 19: 88-101.
- Tan ET, Johnson RH, Lambie DG, et al. Alcoholic vagal neuropathy: recovery following prolonged abstinence.J NeurolNeurosurg Psychiatry. 1984; 47:1335-7.
- Chapman CR, Oka S, Bradshaw DH, et al. Phasic pupil dilation response to noxious stimulation in normal volunteers: relationship to brain evoked potentials and pain report.Psychophysiology. 1999;36: 44-52.
- Lee H, Kim Y, Park J. Pupil cycle time and contrast sensitivity in type II diabetes mellitus patients: a pilot study.Indian J Ophthalmol. 2011; 59: 201-5.
- Tsiptsios D, Fotiou DF, Haidich AB, et al. Evaluation of pupil mobility in patients with myasthenia gravis.ElectromyogrClinNeurophysiol. 2008; 48: 209-18.
- Kono K, Miyao M, Ishihara S, et al. Hypersensitivity in the pupil dilation response to a cholinergic antagonist in patients with Alzheimer’s disease and Down’s syndrome.Nihon Ronen IgakkaiZasshi. 1996;33:829-34.
- Fotiou DF, Brozou CG, Haidich AB, et al.Pupil reaction to light in Alzheimer’s disease: evaluation of pupil size changes and mobility.Aging ClinExp Res. 2007; 19: 364-71.
- Fountoulakis KN, Siamouli M, Kaprinis G, et al. Changes in the pupil reflex arc in depressive patients.J Affect Disord. 2005; 87: 341-2.
- Ionescu S. Psychophysiology of mental deficiency: evaluation of studies involving the recording of autonomic indices. PsychiatrEnfant. 1985;28:39-134.
- Richman JE, McAndrew KG, Decker D, Mullaney SC. An evaluation of pupil size standards used by police officers for detecting drug impairment.Optometry. 2004; 75: 175-82.
- Yanagawa Y, Miyazaki M, Sakamoto T. Relationship between abnormal pupillary reactivity and the outcome of a psychotropic drugAm J EmergMed. 2010; 28: 703-7.
- Vermersch, P, Dufourd-Delalande, S, Defoort-Dhellemmes S, et al. Tonic pupils in Sjogren’s syndrome, Rev Neurol 2005; 161: 963-6.
- Surakka J, Ruutiainen J, Romberg A, et al. Pupillary function in early multiple sclerosis.ClinAuton Res. 2008; 18: 150-4.
- Armstrong RA. Visual signs and symptoms of Parkinson’s disease.ClinExpOptom. 2008; 91: 129-38.
- Tokuda Y, Nakazato N, Stein GH. Pupillary evaluation for differential diagnosis of coma. Postgrad Med J 2003;79:49–51.
- Clusmann H, Schaller C, Schramm J. Fixed and dilated pupils after trauma, stroke, and previous intracranial surgery: management and outcome. J NeurolNeurosurg Psychiatry 2001; 71: 175-81.
- Chesnut RM, Ghajar J, Maas AI, et al. Management and prognosis of severe traumatic brain injury. Part II: Early indicators of prognosis in severe traumatic brain injury. Brain Trauma Foundation, American Association of Neurological Surgeons, Joint section on neurotrauma and critical care. 2000;41:54.
- Marmarou A, Lu J, Butcher I, et al .Prognostic Value of the Glasgow Coma Scale and Pupil Reactivity in Traumatic Brain Injury Assessed Pre-Hospital and on Enrollment: An IMPACT Analysis. J Neurotrauma, 2007; 24: 270-80.
- Shamim MS, Qadeer M, Murtaza G, et al.Emergency department predictors of tracheostomy in patients with isolated traumatic brain injury requiring emergency cranial decompression.J Neurosurg. 2011; 115 :1007-12.
- Al-Jishi A, Saluja RS, Al-Jehani H, et al. Primary or secondary decompressivecraniectomy: different indication and outcome.Can J Neurol Sci. 2011; 38: 612-20.
- Kim YJ.A systematic review of factors contributing to outcomes in patients with traumatic brain injury.J ClinNurs. 2011 ;20 :1518-32.
- Tien HC, Cunha JRF, Wu SN, et al. Do Trauma Patients with a Glasgow Coma Scale Score of 3 and Bilateral Fixed and Dilated Pupils Have Any Chance of Survival? J Trauma, 2006; 60: 274-8.
- Hoffmann M, Lefering R, Rueger JM, et al. Trauma Registry of the German Society for Trauma Pupil evaluation in addition to Glasgow Coma Scale components in prediction of traumatic brain injury and mortality.Br J Surg. 2012 Jan;99:122-30.
- Franschman G, Peerdeman SM, Andriessen TM, et al. Analysis of Results and Methods–Traumatic Brain Injury (ALARM-TBI) Investigator. Effect of secondary prehospital risk factors on outcome in severe traumatic brain injury in the context of fast access to trauma care.J Trauma. 2011;71:826-32.
- Cooper DJ, Rosenfeld JV, Murray L, et al. Decompressivecraniectomy in diffuse traumatic brain injury.N Engl J Med. 2011; 364: 1493-1502.
- Chi JH.Craniectomy for traumatic brain injury: results from the DECRA trial.Neurosurgery. 2011 ;68 :N19-20.
- Rose D, Meeker M, Bacchetti P, et al. Evaluation of the portable infrared pupillometer. Neurosurgery 2005; 57: 198-203.
- Taylor WR, Chen JW, Meltzer H, et al. Quantitative pupillometry, a new technology: normative data and preliminary observations in patients with acute head injury. J Neurosurgery, 2003; 98: 205-13.
- neuroptics.org./pi_faq_110721.pdf
- Surakka J, Ruutiainen J, Romberg A, et al. Pupillary function in early multiple sclerosis. ClinAuton Res. 2008;18 :150-4.
- Matouskova O, Slanar O, Chytil L, et al. Pupillometry in healthy volunteers as a biomarker of tramadol efficacy.J ClinPharmTher. 2011 ;36:513-7.
- Larson MD, Berry PD. Supraspinal pupillary effects of intravenous and epidural fentanyl during isofluraneanesthesia.RegAnesth Pain Med. 2000; 25:60-6.
- Charier D,lZantour D, Pichot V, et al. Evaluation of Analgesia During General Anesthesia: Pupillometry Versus Heart Rate Variability. ASA meeting abstracts 2012; A187.
- Constant I, Nghe MC, Boudet L, et al. Reflex pupillary dilatation in response to skin incision and alfentanil in children anaesthetized with sevoflurane: a more sensitive measure of noxious stimulation than the commonly used variables. Br J Anaesth. 2006; 96:614-9.
- Bourgeois E, Sabourdin N, Louvet N, et al.Minimal alveolar concentration of sevoflurane inhibiting the reflex pupillary dilatation after noxious stimulation in children and young adults.Br J Anaesth. 2012;108:648-54.
- Merlin S, Larson D, Berry PD, et al. Latenchy of papillary reflex during general anesthesia. J ApplPhysiol 2004; 97:725-30.
- Guglielminotti J, Mentré F, Gaillard J, et al. Assessment of pain during labor with pupillometry: a prospective observational study. Anesth Analg. 2013;116:1057-62.
- Kantor E, Montravers P, Longrois D, et al. Pain assessment in the postanaesthesia care unit using pupillometry: A cross-sectional study after standard anaesthetic care.Eur J Anaesthesiol. 2014 ;31:91-7.
- http://clinicaltrials.gov/show/NCT01685645
- Behrends M, Niemann CU, Larson MD. Infrared pupillometry to detect the light reflex during cardiopulmonary resuscitation: A case series.Resuscitation. 2012 ;83 :1223-8.
- Breckwoldt J, Arntz HR. Infrared pupillometry during cardiopulmonary resuscitation for prognostication—A new tool on the horizon? Resuscitation.2012 ;83:1181-2.
- Okada K, Ohde S, Otani N, et al.Prediction protocol for neurological outcome for survivors of out-of-hospital cardiac arrest treated with targeted temperature management.2012;83:734-9.
- Suys T, Bouzat P, Marques-Vidal P, et al. Automated Quantitative Pupillometry for the Prognostication of Coma After Cardiac Arrest. NeuroCrit. Care 2014; [Epub ahead of print]
- Wijdicks EF, Hijdra A, Young GB, et al. Practice parameter: prediction of outcome in comatose survivors after cardiopulmonary resuscitation (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2006;67:203-10.
- Denny JC, Arndt FV, Dupont WD, et al. Increased hospital mortality in patients with bedside hippus. J Med. 2008; 121:239-45.
- Chaudhuri K, Malhma GM, Rosenfield JV. Survival of patients with coma and bilateral fixed pupils. Injury 2009 ;40 :28-32.
- Schnell D, Arnaud L, Lemial V, et al. Pupillary hippus in nonconvulsive status epilepticus. Epileptic Disord. 2012; 14:310-2.
- Legriel S, Azoulay E, Resche-Rigon M, et al. Functional outcome after convulsive status epilepticus. Crit Care Med 2010; 38: 2295-303.
- Chen JW, Gombart ZJ, Rogers S, et al. Pupillary reactivity as an early indicator of increased intracranial pressure: The introduction of the neurological pupil index. SurgNeurolInt 2011; 2:82.
- Kuo JR, Lo CJ, Lo CL, et al. Prognostic predictor of outcome in an operative series in trauma brain injury. J Formos Med Assoc 2011; 110:258-64.
- Hautin E, Cour M, Illinger M, et al. Intérêt de l’ examen pupillair eautomatiséen réa Réanimation 2010; 19 (Suppl 1) :S40.
- Bader MK. Gizmos and Gadgets for the Neuroscience Intensive Care Unit. J NeurosciNurs. 2006; 38:248-60.
- Paulus J, Roquilly A, Beloeil H, et al. Pupillary reflex measurement predicts insufficient analgesia before endotracheal suction in critically ill patients. Crit. Care 2013; 17:R161.
- Payen JF, Isnardon S, Lavolaine J, et al. Pupillometry in anesthesia and critical care.Ann Fr AnesthReanim. 2012; 31:155-9.
- Κοντογούνης Γεώργιος: Μελετη των διαγνωστικών δυνατοτήτων της κορημετρίας σε νευρολογικούς ασθενείς στη μονάδα εντατικής θεραπείας. Ιατρική σχολή ΑΠΘ. Διδακτορική διατριβή (τρέχουσα). Επιβλέπων: καθ. Τάσκος Νικόλαος
- Murphy P, O’Connel R, O’Sullivan M, et al. Pupil diameter covaries with bold activity in human locus ceruleus. Hum Brain Mapp.2014;35:4140-54.
Author Disclosures:
Authors Aslanidis Th., Kontogounis G. have no conflicts of interest or financial ties to disclose.
Corresponding author:
Theodoros Aslanidis,
4 Doridos Street 54633,
Thessaloniki, Greece,
tel.: +306972477166,
e-mail: